Products
-

ABMEDICA
abmedica.fr
Not available in Canada - in regulatory registration processABMEDICA
DB2C was founded in 1992 as a manufacturer and supplier of premium reusable surgical instruments for laparoscopy. The engineering and the quality of the creative products position DB2C as a leading supplier in the French surgery market.
abmedica acquires DB2C in 2009 and expands its activities in the distribution of devices for highly innovative medical devices for laparoscopy and endovascular treatments.
DB2C / abmedica specializes in the fields of laparoscopic surgery, cardiac surgery and interventional neuroradiology.
***In regulatory registration process
Website -

BAXTER
advancedsurgery.baxter.comBAXTER - Advanced Surgery
Baxter's portfolio of products and programs are foundational to the cadence of surgery, just as surgeons, nurses, and administrators are essential to the art of healing. Clinically-proven products allow surgical teams freedom to innovate and think creatively without restraint in the operating room. Best-in-class programs provide a holistic approach to optimizing patient care.
Website -

DORNIER
dornier.comDORNIER
With world's bestselling lithotripter and Europe’s market-leading laser Solvo, Dornier MedTech, headquartered in Munich, Germany, is a medical device company known for its pioneering technologies and revolutionary therapies in urology. Thanks to its deep-rooted spirit of entrepreneurship, excellence and innovation, Dornier thrives while developing some of the highest quality urology products on the market today.
Website -

EQUIPMENT MAINTENANCE PROGRAM
EQUIPMENT MAINTENANCE PROGRAM
The founder and Liaison Medical management team have been in the industry for over 25 years and were part of solid companies’ executive teams. Their experience and knowledge gave them the opportunity to meet skilled partners that have been providing complete maintenance solutions for all major brands of equipment since 1990.
Liaison Medical’s willingness is to partner with you addressing your healthcare system growth, quality, and operational excellence. Our EQUIPMENT FOLLOWER MAINTENANCE (EFM) program will help you meet your business objectives.
Our priority is providing quality workmanship, superior parts, rapid turn-around time and reducing the number of events so our customers can focus on what’s more important…the patient.
-

Evergen Bio
evergenbio.comEvergen Bio
A world-class Contract Development and Manufacturing Organization (CDMO) that partners with innovative companies to accelerate the design, development, and manufacturing of regenerative biomaterial solutions that help patients heal. Evergen, formerly known as RTI Surgical. Our new name reflects our renewed commitment to accelerating the development of cutting-edge, patient-centric regenerative medicine solutions.
Website -
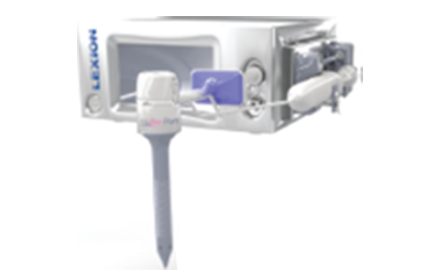
LEXION
lexionmedical.comLEXION
LEXION Medical is a world leader in the design and manufacturing of advanced technologies for gas conditioning in minimally invasive surgical procedures. Our goal is to provide innovative technologies that help patients return to their normal, productive lives faster. Appropriately conditioning the gas before it enters the body and when it leaves the patient can dramatically help patients, surgeons, and make operating rooms more efficient with improved clinical outcomes.
Website -

NUVASIVE
NUVASIVE
NuVasive® is one of the fastest growing, comprehensive spine companies in the world. NuVasive is committed to providing surgeons a portfolio that minimizes disruption and delivers the best possible patient outcomes, including the industry leading XLIF® procedure.
-

SPINEVISION
spinevision.netSPINEVISION
SpineVision® is a privately-owned integrated spinal technology company focused on the development and marketing of implants and instrumentation for spinal treatment.
Since its foundation in 1999, the company has designed innovative products which offer key advantages to surgeons and benefits to patients. SpineVision’s current products offer solutions for approximately 90% of spinal pathologies i.e. lumbar degenerative disc diseases, deformities, cervical disorders, trauma and tumors. With its new generation products Flex+2 and LUMIS SpineVision® is the only manufacturer offering a dynamic stabilization system which can be used in a percutaneous approach.
The Company is headquartered in Antony (south of Paris), France, and has subsidiaries in the USA, Italy, Belgium and UK.
Website -
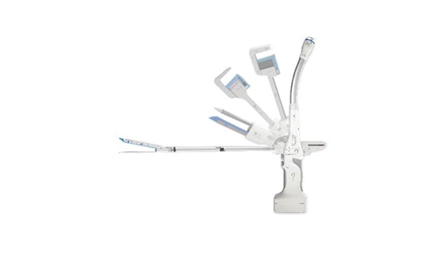
INTOCARE
intocare.comINTOCARE
Intocare Medical Technology Co., Ltd, founded in 2015, is a Chinese high-tech corporation focusing on surgical instruments.
Excellent product comes from the profound insight into demand and constant innovation. “Insight and Innovation to Caring”- this is the meaning of Intocare.
Intocare is currently located in Nanopolis Suzhou, Suzhou Singapore Industrial Park. It is an industrial integrated community, in which major R&D engineering platform, high-end innovation team, industrial development service institution and promotive organization for international industry are located.
Website -
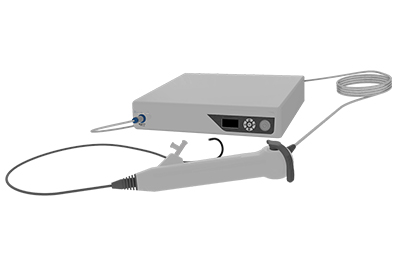
DISPOSABLE ENDOSCOPE
About
Liaison Medical is a Canadian medical distributor specializing in the delivery of innovative medical technologies to hospitals. Based upon our extensive experience, we are the liaison between Manufacturers and hospitals for the Operation Room and Endoscopy departments.
Contact Us

Québec
Phone: (450) 985-1696
Fax: (450) 985-1696
Email: info@liaisonmedical.ca
